Helium diffusing through mylar foils will contaminate the DME in the E614
wire chambers. The diffusion rate and its variability are studied, and
the implications for E614 are examined. It was determined that the
diffusion is low enough and (more importantly) stable enough that DME
buffer zones will not be needed.
The attachments are:
1) hereport.ps The report in Postscript format.
2) leaktst.gif Schematic of the experimental apparatus.
(Also embedded in both report formats.)
3) hereport.html The report in HTML format.
For the TRIUMFhttp://www.triumf.ca">TRIUMF> E614http://www.phys.ualberta.ca/~rodning/E614/">E614> Collaboration.
November 12, 1997
This document is also available in postscript format.
The proposed detector for the E614 project contains chambers filled with helium gas and wire chambers filled with DME, separated by 6 micron, double-sided aluminized mylar foils. This experiment was designed to determine how quickly the helium diffuses through the foil into the DME chamber, and more importantly, whether this diffusion rate is enough to cause significant helium contamination of the DME. Changes of the gas mixture in the wire chambers alters drift speeds, which ultimately affects the spatial resolution of the detector.
The diffusion rate per unit area of foil was found to be about 3.6E-6 cc s-1 Torr-1 m-2 (+/- 30%) on average, which implies a helium diffusion rate of 2.5E-4 cc/s (0.015 cc/min) into the wire chambers in the detector. The wire chambers are to be flushed with DME at a proposed rate of 20 cc/min (+/- 10%), which means there will be about 0.1% helium contamination in the DME with fluctuations of +/- 0.01% contamination. Drift-time calculations performed by Renee Poutissou using the GARFIELD simulation package show that the effects of these fluctuations, and even the effects of the contamination level itself, are negligible.
The detector to be used in the E614 experiment will contain chambers filled with helium gas and wire chambers filled with dimethyl ether (DME), separated by 6 micron, double-sided aluminized mylar foils. Helium can diffuse through the aluminized mylar, contaminating the DME and affecting measurements (by altering electron drift times, for example). This experiment was designed to determine how quickly helium diffuses through the foil and whether or not it will significantly contaminate the DME.
The HERMES project has made similar measurements for 25-micron foils, finding a diffusion rate of about 100 cc day-1 atm-1 m-2 (that is, 100 cc diffuse through each square metre of the foil per day for each 1 atm difference in pressure across the foil), or 1.5E-6 cc s-1 Torr-1 m-2. The diffusion rate for 6 micron foil was expected to be only slightly higher than this.
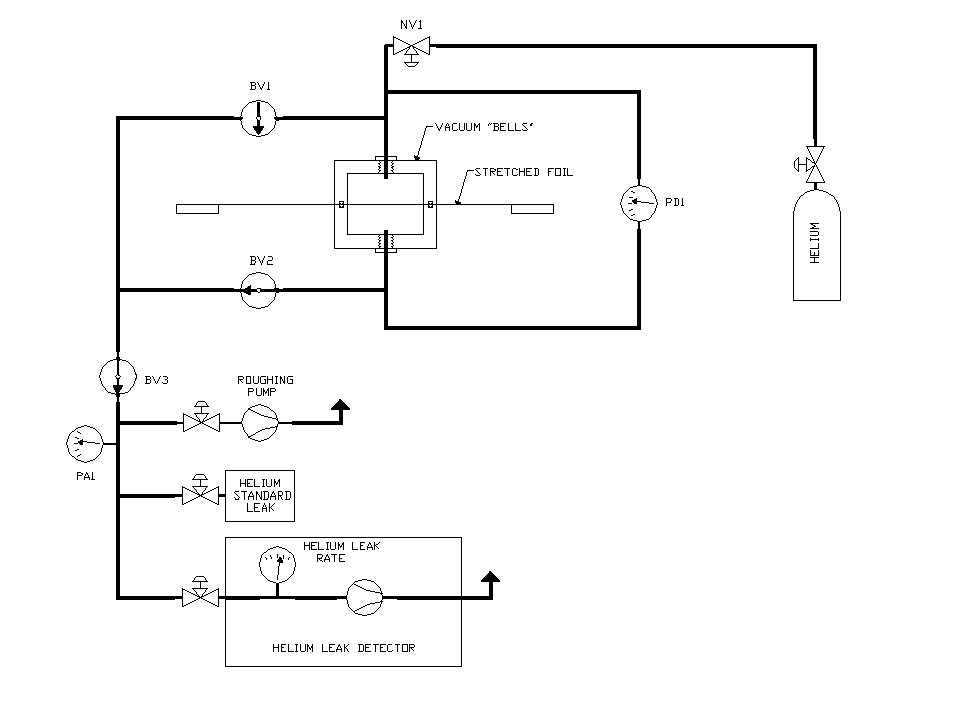
Figure 1: Experiment Schematic. NV1 and similar symbols are
needle (throttle) valves. BV1, BV2, and BV3 are ball valves, used to isolate parts
of the system. The vacuum "bells" are aluminum cups with o-ring
seals. PD1 is a differential pressure gauge, while PA1 is an absolute pressure
gauge. All elements are connected with Polyflow tubing.
The heart of the experiment consisted of aluminum vacuum "bells" (made by Pierre Amaudruz) with rubber o-ring seals, placed on opposite sides of a stretched foil. The bells were connected via Polyflow tubing to a differential pressure gauge, as well as to vacuum pumps and a helium leak detector. One bell was also connected to a helium tank, through a Polyflow needle valve. Ball valves were used to isolate various parts of the system, and throttle valves were used to control gas flow (eg. from the helium tank). Connections were made using both Polyflow and Swagelok fittings. See figure 1 for the schematic.
Several sections of mylar foil were stretched using a standard vacuum stretching frame, and transferred to mounting frames; the mounted foils were referred to as "test plates". Each foil was examined against a strong backlight to look for "spots" (points where light shines through the foil more strongly, probably due to the absence of aluminum on one side of the foil at that spot) and a rough visual estimate of the "spot density" was made (1 = very few spots, 5 = very many spots) for regions of the foil to be tested.
A "bell" was positioned on each side of the stretched foil; the system was connected to a helium leak detector and the absolute pressure of the entire system was lowered to a few millitorr. The leak detector was isolated (using ball valve BV3 in figure 1), and calibrated using the helium standard leak; BV3 was then reopened so that measurements could be taken.
The "helium" side of the foil was isolated during the actual test (using BV1 in figure 1); helium was drawn into the bell on that side of the foil until the differential pressure (measured with PD1) across the foil was about 1 Torr. The rate at which the helium diffused through to the "detector" side was measured using the helium leak detector.
Multiple measurements were made at some foil positions to test the reproducibility of the results. At many positions, more helium was added to the "helium"-side bell to raise the pressure across the foil to about 2 Torr, and extra measurements were taken to test the diffusion rate for linearity. Some drifting of the calibration gain was observed (the detector reading of the controlled leak grew steadily weaker over time, due to a loss of detector sensitivity - see "Sources of Error") during the early part of the experiment, and so the calibration was repeated frequently.
The measurements taken using the first two plates were discarded. Problems with the leak detector (including the problems with the calibration gain, as mentioned above) made measurements taken using those plates unreliable. The problems were later corrected, and measurements taken after the corrections are considered accurate (to within 30%; see "Results"). The measurements were repeated using a 25 micron, double-sided aluminized mylar foil, to make sure that the results of this experiment were consistent with those arrived at by HERMES.
Each measured rate of diffusion through 6-micron foil was divided by the differential pressure (across the foil) to obtain a rate per unit pressure. The mean of these rates, taken over every "reliable" measurement obtained (that is, those measurements taken after the problems with calibration gain were corrected), was found to be 1.1E-8 cc s-1 Torr-1 through the section of foil contained by the vacuum bells, with a standard deviation of 2E-9 cc s-1 Torr-1 (about 20%). This represents the average of 17 data points.
The area of this section of foil (based on the diameter of the inside edge of the o-ring groove in the bell) was 3.50E-3 m2, giving a diffusion rate per unit area of 3.6E-6 cc s-1 Torr-1 m-2 (or 240 cc day-1 atm-1 m-2). (This value for the diffusion rate through 6 micron foil is within an order of magnitude of the HERMES measurement for 25 micron foil, as expected. Though the HERMES measurements were taken using a 25 micron foil, it was expected that the diffusion rate through 6 micron foil should be similar, at least to within an order of magnitude.)
The measured diffusion rate (per unit pressure) differed significantly between foils, between positions on the same foil, and even between measurements at the same position on a given foil. Variations were as large as 30%.
The proposed E614 detector will use circular 6-micron foils of radius 0.169 m, with helium partial pressure at 1 atm on one side, and 0 atm on the other (except for whatever diffuses through). For these conditions, helium will diffuse through the foil into the DME at a rate of (3.6E-6 cc s-1 Torr-1 m-2)(760 Torr)(0.169 m)2pi = 2.5E-4 cc/s, or 0.015 cc/min. The DME will be flushing the wire chamber at a proposed nominal rate of 20 cc/min, resulting in about a 0.1% helium contamination of the DME. The DME flow will be stable to +/- 10%, which means the helium concentration in the DME will be stable to +/- 0.01%.
According to GARFIELD simulations by Renee Poutissou, a 1% change in helium levels in the DME will cause a 1% change in electron drift time, in a linear relationship. Since the contribution to drift time instability is on the order of 0.01%, special measures (such as DME buffer chambers) need not be taken to protect the DME-filled wire chambers from helium contamination.
It should be noted that diffusion through the foil is not the only possible source of helium contamination in the DME chambers. Even a small pinhole in the foil or the chamber seals can result in contamination which is orders of magnitude greater than any contamination by diffusion. It is vital, therefore, that the chambers be completely leak-tight, and that they be tested for leaks before being put into operation.
Individual measurements taken in regions of high spot density and in regions of low spot density were compared, and no measurable difference was detected above measurement error (more significant changes were often observed between separate measurements at the same foil position than between some of the measurements at regions of different spot density).
The mean diffusion rate through a 25 micron foil was measured (through the same procedure as above) to be 7.8E-9 cc s-1 Torr-1 through the section of foil contained by the vacuum bells, and the standard deviation was 4.2E-9 cc s-1 Torr-1. This converts to a diffusion rate per unit area of 2.6E-6 cc s-1 Torr-1 m-2, or 170 cc day-1 atm-1 m-2, which is within a factor of 2 of the HERMES measurement of 100 cc day-1 atm-1 m-2. Other groups have run similar experiments, and have reported (through private communications) that the diffusion rate through a given foil is often different by a factor of 2 or 3 from the diffusion rate through another foil of the same thickness.
The helium leak detector was problematic throughout the experiment. The most notable problem was a total loss of sensitivity, which recurred on an almost regular basis. The sensitivity (as measured by testing the leak detector with the controlled leak) would sometimes decrease over time, particularly during the early part of the experiment. The sensitivity appeared very stable during the last part of the experiment, however, when most of the data used was taken. The leak detector was re-calibrated frequently (once every one or two measurements) to ensure the stability of the calibration and reduce this source of error.
The diffusion rates being measures were at the very bottom end of the helium leak detector's scale, and as such the experiment was highly susceptible to small leaks, fluctuations in voltages within the detector itself, etc. Small air leaks in the apparatus were detected but not located; to correct for these leaks, the detector reading before the "helium"-side bell was isolated was subtracted from the reading after the "helium"-side bell was filled with helium.
Helium and other gases are able to diffuse through the Polyflow tubing used in the experiment, and this may have had a small effect on the readings. The most likely place for this effect to cause problems is in the long tube leading to the helium tank, as it would lower the concentration of helium in the tube. For this reason, this tube was flushed with helium each morning while the experiment was in progress.
Occasionally, diffusion rates 10 or 100 times higher than expected were measured. Examination of the foil revealed that these rates were due to small (and sometimes larger) holes in the foil, created when the cups were positioned on the foil or when the foil was damaged in some other way. These holes were often very small, but they allowed much more helium through than diffusion alone. Upon detection of a hole in the foil, measurements for that foil location were discarded.
Report by Robert
MacDonald.
Last modified: December 10, 1997.
Figure 1 generated with AutoCAD by
Robert Openshaw.